Introduction
- Clinical laboratory examination of urine provides important information about both renal diseases and systemic diseases that affect the urinary system.
- Urine analysis helps detect disorders related to the kidneys, ureters, urinary bladder, and urethra.
- Both structural abnormalities and functional disturbances of the kidneys and lower urinary tract can be identified through urine examination.
- Laboratory examination of urine also provides sequential information about the nature of disease, possible cause, and prognosis.
- Careful urine analysis helps narrow the clinical differential diagnosis of many renal disorders.
- It is one of the simplest and most valuable laboratory investigations used in routine clinical practice.
- Proper collection, examination, and interpretation of urine remain essential for accurate diagnosis in clinical medicine.
Normal Constituents of Urine in a 24-Hour Sample
A 24-hour urine sample gives a more accurate picture of the total amount of substances excreted by the body in one day. It is commonly used for biochemical estimation because many urinary constituents vary during different times of the day.
| Constituent | Normal Amount in 24 Hours |
|---|---|
| Water | 1300 – 1600 mL |
| Total Nitrogen (Kjeldahl) | 10 – 17 g |
| Urea | 20 – 35 g |
| Uric Acid | 0.1 – 2.0 g |
| Creatinine | 1.0 – 1.5 g |
| Amino Acids (free and combined) | 1.5 g |
| Glucose | 0.1 g |
| Non-glucose reducing substances | 1.0 g |
| Citric Acid | 0.3 g |
| Ascorbic Acid | 0.025 g |
| Oxalate | 0.015 g |
| Acetone Bodies | 0.01 g |
| Total Sulfur | 1.0 g |
| Phosphate (as P) | 0.4 – 2.2 g |
| Sodium | 3 – 6 g |
| Potassium | 2 – 4 g |
| Calcium | 0.2 g |
Explanation of Important Constituents
Water
- Water forms the major part of urine.
- It helps dissolve waste materials and remove them from the body.
Urea
- Urea is the major nitrogenous waste product formed in the liver.
- It is produced from protein metabolism.
Uric Acid
- Uric acid is formed from purine metabolism.
- It is excreted in small amounts under normal conditions.
Creatinine
- Creatinine is produced from muscle metabolism.
- It is excreted at a relatively constant rate and is useful for kidney function assessment.
Electrolytes
- Sodium, potassium, calcium, phosphate, and chloride are normally excreted in urine.
- Their amount depends on diet, hormones, and kidney function.
Urine Collection and Preservation
Proper urine collection and preservation are essential for valid laboratory reporting.
Types of Urine Collection
1. Random Specimen
- Random urine specimen is the most commonly collected sample.
- It can be collected at any time during the day.
- It is easy to obtain and immediately available.
Common Uses
- Glucose detection
- Ketone bodies
- Bile pigments
- Blood pigments
2. Midstream First Morning Specimen
- This is the preferred sample for routine urinalysis.
- Overnight urine remains in the bladder for several hours, so analytes become concentrated.
- It gives better laboratory accuracy.
Method of Collection
- The patient should discard the first portion of urine.
- The middle portion of urine is collected in a clean container.
- Usually 15–20 mL urine is sufficient.
Advantages
- Concentrated sample
- Less contamination
- Better for microscopic examination
Storage
- Urine should be examined immediately.
- If delayed, it should be refrigerated for one or two days.
3. 24-Hour Urine Collection
- 24-hour urine collection is required when substances show daily variation in excretion.
Used For Estimation Of
- Creatinine
- Urea nitrogen
- Calcium
- Proteins
- Glucose
- Sodium
- Potassium
- Hormones such as catecholamines and hydroxysteroids
Method of 24-Hour Collection
- Start collection at 6 AM.
- Ask the patient to empty the bladder and discard the first sample.
- Collect all urine passed after that into a container.
- Continue collection for the next 24 hours.
- Include the urine passed exactly at 6 AM next morning as the final sample.
Preservatives Used for 24-Hour Urine Collection
Common Preservatives
- 50 mL of 2N HCl
- 10 mL concentrated HCl
- Thymol crystals
- 5 mL thymol solution in isopropanol
These preservatives prevent bacterial growth and chemical changes in urine.
Routine Examination of Urine
Routine urine examination is usually divided into three sections:
a. Physical Examination
- Color
- Volume
- Appearance
- Odor
- Specific gravity
b. Chemical Examination
- Protein
- Glucose
- Ketone bodies
- Bile salts
- Blood
c. Microscopic Examination
- Cells
- Crystals
- Casts
- Microorganisms
Physical Examination
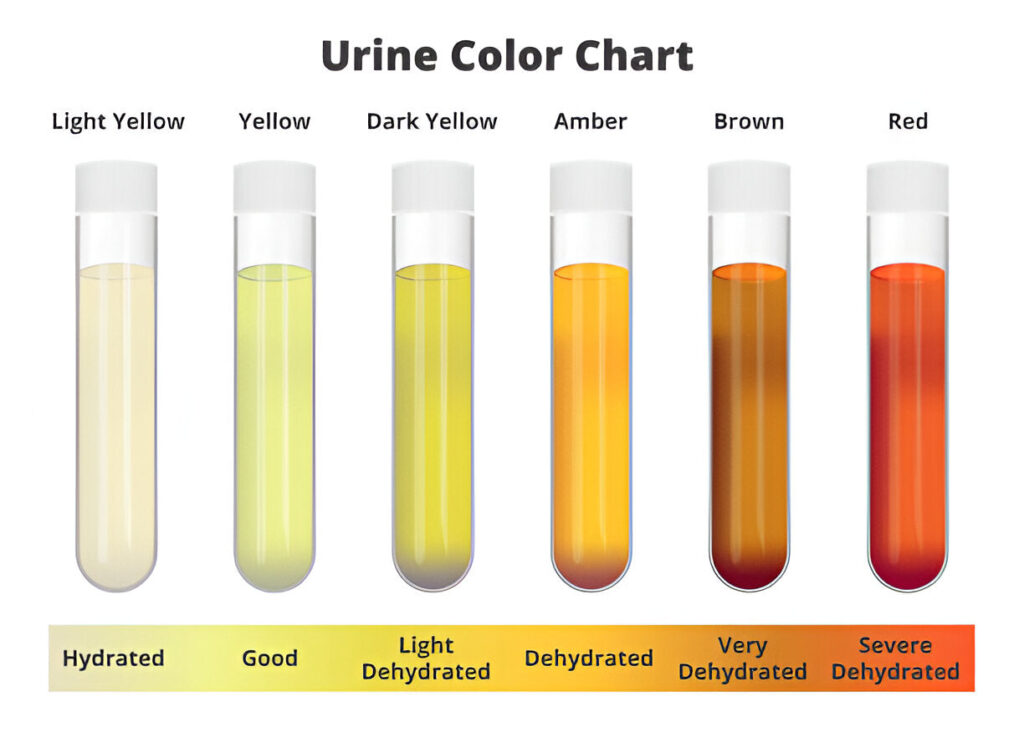
1. Color of Urine
- Urine color depends mainly on its degree of concentration and the pigments present in urine.
- Normal urine is usually pale yellow because of the pigment Urochrome.
- Dilute urine appears lighter, while concentrated urine appears darker.
- Abnormal urine color may indicate presence of blood pigments, bile pigments, metabolic products, or disease.
Normal and Abnormal Urine Colors
| Color | Substance Present | Clinical Condition |
|---|---|---|
| Pale yellow | Urochrome | Normal healthy urine |
| Yellow-orange | Urobilin | Dehydration, jaundice |
| Red | RBC / Hemoglobin | Hematuria, hemoglobinuria |
| Red-brown | Myoglobin / Porphyrin | Myoglobinuria, porphyrinuria |
| Yellow-green | Bilirubin / Biliverdin | Jaundice |
| Brown-black | Homogentisic acid / Methemoglobin / Porphyrin | Alkaptonuria |
| Milky white | Chyle | Chyluria |
Important Abnormal Colors
Red Urine
- Red urine usually indicates blood pigments.
Causes
- Red blood cells in urine (hematuria)
- Hemoglobin in urine (hemoglobinuria)
- Menstrual contamination
Clinical Importance
- May indicate urinary tract bleeding, kidney disease, or hemolysis.
Red-Brown Urine
- This occurs when muscle pigments or porphyrins are present.
Causes
- Myoglobinuria
- Porphyrinuria
Clinical Importance
- Seen in muscle injury or certain metabolic disorders.
Yellow-Orange Urine
- Dark yellow urine indicates concentrated urine.
Causes
- Dehydration
- Jaundice
Yellow-Green Urine
- Occurs due to bile pigments.
Causes
- Bilirubin
- Biliverdin
Clinical Importance
- Suggests liver disease or obstructive jaundice.
Brown-Black Urine
- Urine may become dark on standing.
Causes
- Homogentisic acid
- Methemoglobin
- Porphyrins
Clinical Importance
- Seen in Alkaptonuria
Milky White Urine
- Urine becomes cloudy white when fat is present.
Causes
- Chyle
Clinical Importance
- Seen in lymphatic obstruction or chyluria.
2. Volume of Urine
- Urine volume depends on:
- Fluid intake
- Solutes excreted such as sodium and urea
- Loss of water through skin, lungs, and bowel
- Cardiovascular status
- Kidney function
- Normally, a healthy adult excretes 1000 to 2500 mL urine per day depending on fluid intake and environmental conditions.

Abnormal Changes in Urine Volume
Polyuria
- Polyuria means urine output greater than 2.5 liters per day.
- It occurs when the kidneys excrete excessive water.
Causes of Polyuria
- Diabetes Mellitus
- Diabetes Insipidus
- Later stages of chronic renal failure
- Use of diuretic drugs
Clinical Importance
- Persistent polyuria may indicate endocrine or renal disease.
Oliguria
- Oliguria means urine output less than 300 mL per day.
- It usually indicates reduced kidney filtration or fluid loss.
Causes of Oliguria
- Fever
- Acute nephritis
- Early chronic glomerulonephritis
- Diarrhea
- Cardiac failure
Clinical Importance
- Oliguria is often an early warning sign of dehydration or kidney injury.
Anuria
- Anuria means complete absence of urine output for 24 hours or extremely minimal urine formation.
Causes of Anuria
- Shock
- Acute tubular necrosis
- Mercury poisoning
- Incompatible blood transfusion
Clinical Importance
- Anuria is a medical emergency and requires immediate evaluation.
3. Appearance of Urine
- Freshly passed normal urine is usually clear and transparent.
- Normal urine may become slightly cloudy on standing because crystals can form.
- Phosphate crystals
- Urate crystals
- Oxalate crystals
Abnormal Turbidity
Urine becomes abnormally turbid due to:
- White blood cells
- Red blood cells
- Bacteria
- Pus cells
Clinical Importance
- Turbid urine may indicate urinary tract infection or inflammation.
4. Odor of Urine
- Normal urine has a slightly ammoniacal odor due to breakdown of urea.
Abnormal Odors and Their Causes
Fruity Odor
- Fruity smell occurs because of acetone in urine.
Cause
- Ketoacidosis, especially in uncontrolled diabetes
Foul Smell
- Foul odor usually suggests bacterial infection.
Cause
- Urinary tract infection
Mousy Smell
- A characteristic mousy odor is found in certain metabolic disorders.
Cause
- Phenylketonuria
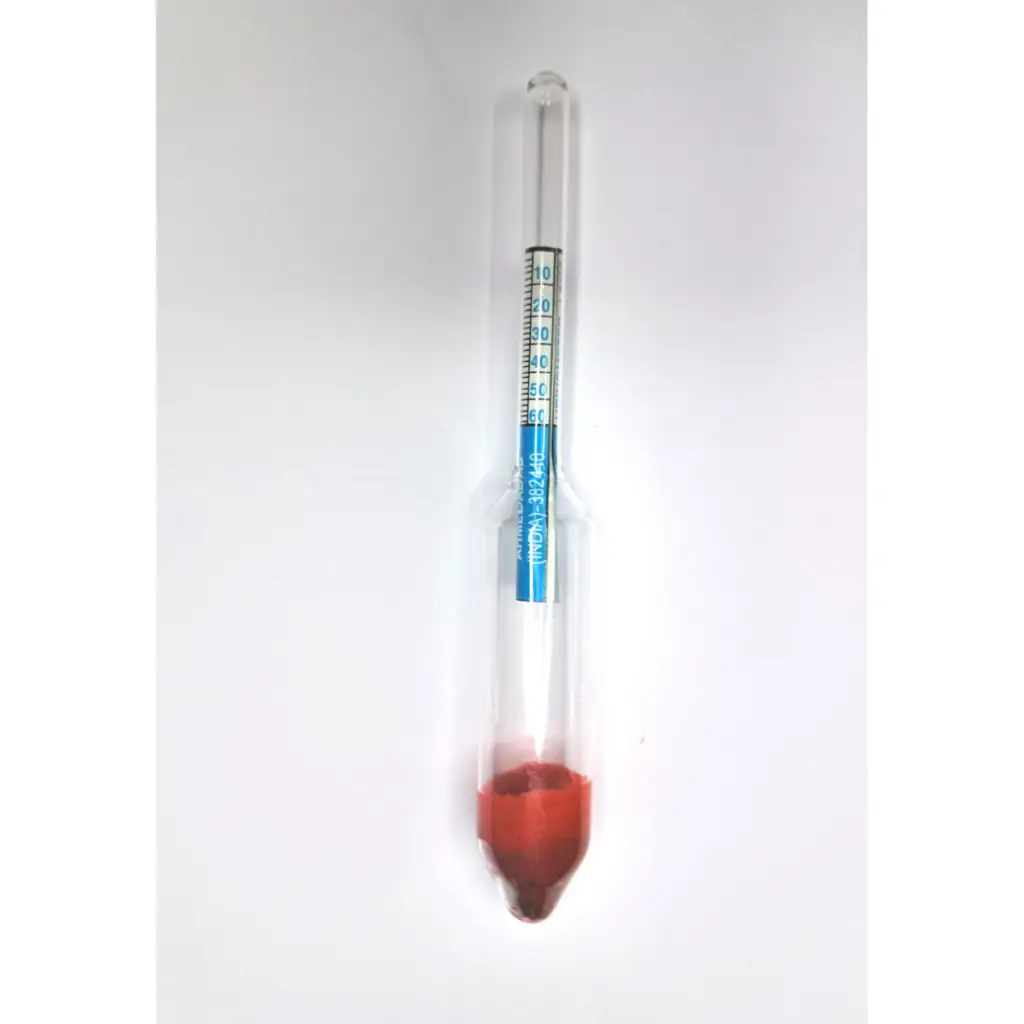
5. Specific Gravity of Urine
- Specific gravity indicates the concentration of dissolved substances in urine.
- It reflects the kidney’s ability to concentrate or dilute urine.
- Specific gravity is directly proportional to the amount of solutes excreted.
Normal Range
- Normal urine specific gravity ranges from 1.012 to 1.024.
- Dilute urine has lower specific gravity.
- Concentrated urine has higher specific gravity.
Instrument Used
- Specific gravity is measured using Urinometer.
High Specific Gravity
- High specific gravity indicates concentrated urine.
Causes
- Restricted water intake
- Dehydration
- Presence of glucose in urine
- Diabetes Mellitus
- Proteinuria
- Adrenal insufficiency
Low Specific Gravity
- Low specific gravity indicates dilute urine.
Causes
- Polyuria
- High fluid intake
- Diabetes Insipidus
- Hypothermia
Fixed Specific Gravity (1.010)
- Specific gravity fixed at 1.010 suggests severe kidney damage.
Clinical Importance
- Seen in chronic renal failure
- Indicates loss of concentrating ability of kidney
Correction of Specific Gravity
Glucose Correction
- For every 1 g/dL glucose, subtract 0.004
Protein Correction
- For every 1 g/dL protein, subtract 0.003
Temperature Correction
- Urinometer is calibrated at 15°C or 20°C
- For every 3°C above calibration, add 0.001
- For every 3°C below calibration, subtract 0.001
6. Reaction of Urine (pH)
- Freshly voided urine is usually slightly acidic.
- It may also be neutral or faintly alkaline.
- On standing, urine becomes alkaline because bacteria convert urea into ammonia.
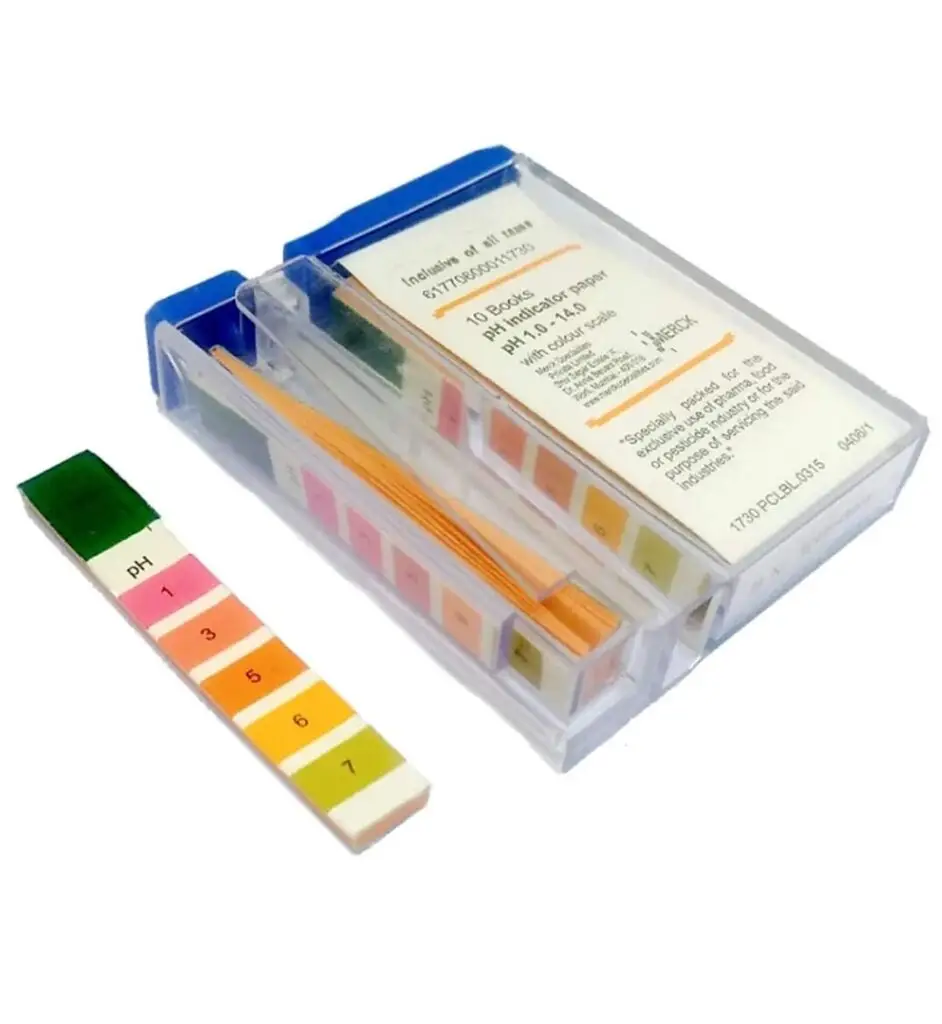
Normal Urine pH
- Normal urine pH ranges from 4.6 to 8.0
Alkaline Tide
- After meals, urine may become alkaline.
- This happens because hydrochloric acid is secreted in the stomach.
- Blood temporarily becomes alkaline.
- This is called alkaline tide.
Effect of Diet on Urine pH
Acidic Urine
- High protein diet
- Low carbohydrate diet
Alkaline Urine
- Diet rich in vegetables and fruits
Causes of Acidic Urine
- High protein diet
- Starvation
- Dehydration
- Diarrhea
- Diabetic ketoacidosis
- Metabolic acidosis
- Respiratory acidosis
Causes of Alkaline Urine
- Vegetable diet
- Vomiting
- Renal tubular acidosis
- Respiratory alkalosis
- Metabolic alkalosis
- Ammonia-producing bacteria
- Chronic renal failure
- Low carbohydrate diet
Chemical Examination
A. Organic Constituents
1. Urea in Urine
Principle (Urease Test)
- Urease enzyme decomposes urea into ammonia and carbon dioxide.
- Ammonia makes the solution alkaline, changing the indicator color from yellow to pink.
Reaction
CO(NH2)2+H2O→2NH3+CO2
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 2 mL urine in one test tube and 2 mL water in another tube | — | — |
| Add one drop of phenol red indicator to both tubes | — | — |
| Add sodium carbonate till pink color appears, then add acetic acid till pink just disappears | — | Slightly acidic medium prepared |
| Add a pinch of soybean powder or horse gram powder | Pink color reappears in urine tube only | Urea present in urine |
Principle (Hypobromite Test)
- Urea reacts with sodium hypobromite and releases nitrogen gas causing effervescence.
CO(NH2)2+3NaOBr+2NaOH→NaBr+N2+Na2CO3+3H2O
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 5 mL urine in a test tube | — | — |
| Add 1 mL alkaline sodium hypobromite | Effervescence occurs | Urea present in urine |
Clinical Interpretation
- Normal urinary urea: 25–30 g/day
Increased Urinary Urea
- High protein diet
- Fever
- Tissue breakdown
Decreased Urinary Urea
- Renal Failure
- Severe liver disease
- Low protein diet
2.Test for Uric Acid
Principle (Phosphotungstic Acid Reduction Test)
Uric acid reduces phosphotungstic acid in alkaline medium to form tungsten blue.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 2 mL urine in a test tube | — | — |
| Add few drops of phosphotungstic acid reagent | — | — |
| Add few drops of 20% sodium carbonate and mix | Deep blue color appears | Uric acid present |
Clinical Interpretation
- Normal urinary uric acid: 250–750 mg/day
Increased Urinary Uric Acid
- High purine diet
- Gout
- Leukemia
- Cortisone or ACTH therapy
Decreased Urinary Uric Acid
- Chronic renal failure
3. Creatinine in Urine
Principle (Jaffe’s Test)
Creatinine reacts with picric acid in alkaline medium to form creatinine picrate, which gives a reddish-orange color.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 3 mL saturated picric acid solution and add 0.5 mL 10% NaOH | — | — |
| Divide into two test tubes | — | — |
| Add urine to one tube and water to the other (control) | Urine tube shows deep reddish-orange color | Creatinine present |
Clinical Interpretation
- Normal urinary creatinine: 1–2 g/day
Increased Urinary Creatinine
- Fever
- Myasthenia Gravis
- Muscular atrophy
- Myositis
- Hyperthyroidism
- Starvation
4. Ammonia in Urine
Principle
Ammonia is liberated when urine is boiled in alkaline medium.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 5 mL urine in a test tube | — | — |
| Add 2% sodium carbonate till alkaline | — | Alkaline medium prepared |
| Boil the solution and hold moist red litmus paper at mouth of tube | Red litmus turns blue | Ammonia present |
Clinical Interpretation
- Normal urinary ammonia: 0.4–1.0 g/day
Increased Urinary Ammonia
- Acidosis
- Diabetes Mellitus
- Starvation
- Hepatic disease
Decreased Urinary Ammonia
- Alkalosis
- Renal failure
- Glomerulonephritis
- Addison’s Disease
B. Inorganic Constituents
1. Chloride
Principle
Chloride reacts with silver nitrate in presence of nitric acid to form white silver chloride precipitate.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 2 mL urine and add 2 drops conc. nitric acid | — | Acidic medium prepared |
| Add 1 mL of 3% silver nitrate | White precipitate forms | Chloride present |
Clinical Interpretation
- Normal: 10–12 g/day
Increased
- Addison’s Disease
- Polyuria
- Diabetes
Decreased
- Sweating
- Diarrhea
- Vomiting
- Burns
2. Inorganic Phosphorus
Principle
Inorganic phosphate reacts with ammonium molybdate in acidic medium to form canary yellow ammonium phosphomolybdate.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 5 mL urine and add few drops conc. nitric acid | — | Acidic medium prepared |
| Add ammonium molybdate and warm | Canary yellow precipitate appears | Phosphate present |
Clinical Interpretation
- Normal: 0.8–1.2 g/day
Increased
- Hyperparathyroidism
Decreased
- Rickets
- Pregnancy
- Nephritis
3. Calcium
Principle
Calcium reacts with potassium oxalate in acidic medium to form calcium oxalate precipitate.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 2 mL urine and add 5 drops acetic acid | — | Acidic medium prepared |
| Add 5 mL potassium oxalate | White precipitate forms | Calcium present |
Clinical Interpretation
- Normal: 0.1–0.3 g/day
Increased
- Hyperparathyroidism
- Hyperthyroidism
- Multiple myeloma
4. Sulfates
Principle
Sulfates react with barium chloride to form barium sulfate precipitate.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 3 mL urine and add few drops conc. HCl | — | Acidic medium prepared |
| Add 1 mL barium chloride | White precipitate forms | Sulfate present |
Clinical Interpretation
- Normal inorganic sulfate: 0.7–1 g/day
Increased
- High protein diet
Decreased
- Renal dysfunction

