Introduction
- Urine examination is an important part of routine laboratory diagnosis because many diseases produce characteristic changes in urine.
- In healthy individuals, urine contains normal constituents in definite amounts and shows normal physical characteristics.
- In pathological conditions, abnormal substances may appear in urine or normal constituents may increase or decrease.
- Changes in appearance, odor, volume, color, pH, specific gravity, and total solids often provide early evidence of disease.
- Detection of abnormal urinary findings helps in diagnosis of renal disorders, metabolic diseases, liver diseases, and systemic conditions.
Physical Characteristics
1. Colour
- Normal urine is pale yellow due to urochrome pigment.
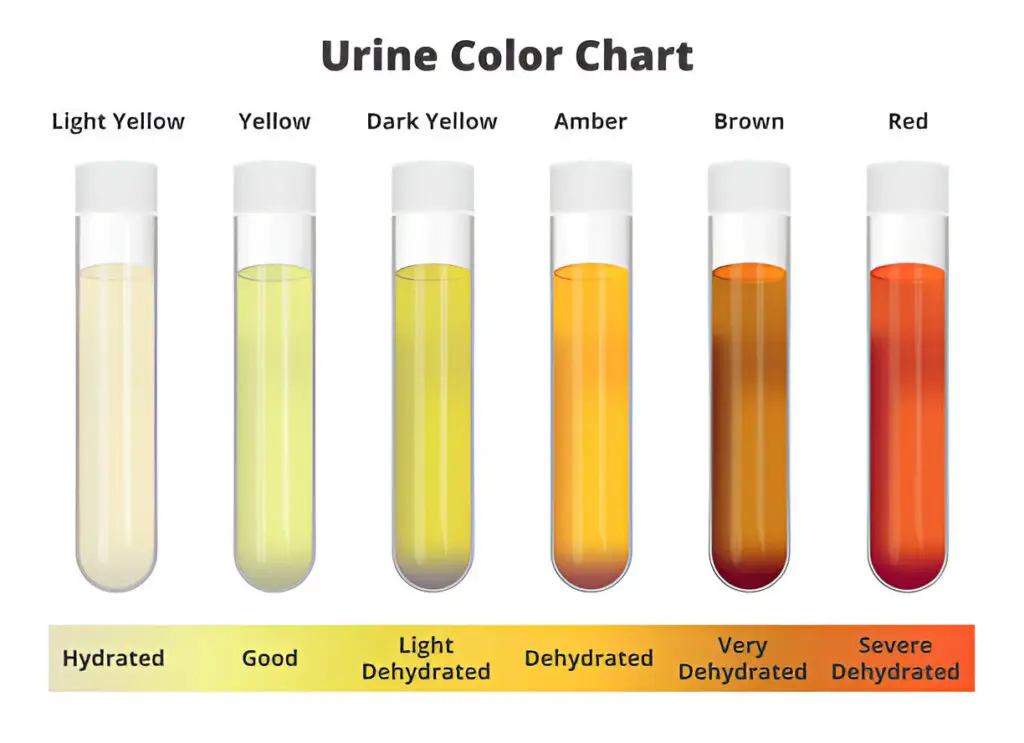
Abnormal Colors
- Smoky brown → blood present
- Yellow → bilirubin present
- Black → melanin present
- Black on standing → Alkaptonuria
- Milky → pus, bacteria, epithelial cells, or lipids
2. Volume
- Normal daily urine output is about 1000–2500 mL.
Increased Volume (Polyuria)
- Seen in:
- Diabetes Mellitus
- Drug administration such as digitalis and salicylates
Decreased Volume (Oliguria)
- Seen in:
- Nephritis
- Fever
- Diarrhea
- Vomiting
Complete Absence (Anuria)
- Seen in:
- Shock
- Acute nephritis
- Incompatible blood transfusion
- Mercury poisoning
- Renal stone
3. Appearance
- Normal urine is clear.
- Urine becomes turbid or opalescent when abnormal substances are present.
Causes of Turbidity
- Proteins
- Pus cells
- Bacteria
- Epithelial cells
- Lipids
4. Odour
- Normal urine has a faint aromatic odor.
- On prolonged standing, urine develops ammoniacal smell because of bacterial decomposition.
Abnormal Odors
- Sweet smell → ketone bodies
- Maple syrup smell → Maple Syrup Urine Disease
5. pH
- Normal urine pH varies from 4.6 to 8.0.
Acidic Urine
- Fever
- Diabetes
- Ketoacidosis
Alkaline Urine
- Alkaline tide after meals
6. Specific Gravity
- Normal specific gravity is 1.015 to 1.025.
Increased Specific Gravity
- Acute nephritis
- Fever
Decreased Specific Gravity
- Diabetes Insipidus
7. Total Solids
- Normal total solids range from 26–80 g/L.
Increased Total Solids
- Occur when abnormal constituents are present in urine.
Clinical Importance
- Physical changes in urine often provide the first clue to disease.
- They help in diagnosis of:
- Kidney disease
- Diabetes
- Liver disorders
- Metabolic disorders
Chemical Constituents of Urine
Reducing Sugars
Benedict’s Reduction Test
Principle
- Reducing sugars in alkaline medium form enediols on heating.
- These enediols reduce cupric ions of Benedict’s reagent to cuprous oxide.
- Cuprous oxide forms colored precipitate depending on sugar concentration.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 5 mL Benedict’s reagent in a test tube | — | — |
| Add 0.5 mL urine (about 8 drops) | — | — |
| Boil for 2 minutes | Green / yellow / orange / red precipitate | Reducing sugar present |
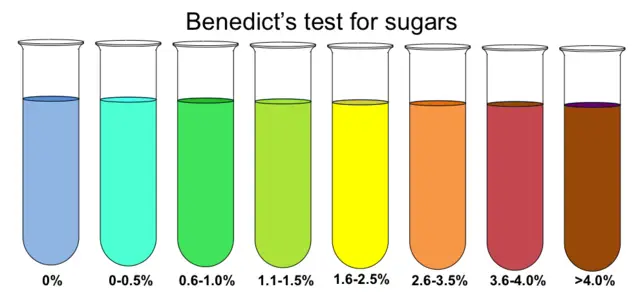
Benedict’s Semiquantitative Interpretation
| Color | Sugar Content (g%) |
|---|---|
| Blue | Nil |
| Green precipitate | 0.1 – 0.5 |
| Yellow precipitate | 0.6 – 1.0 |
| Orange precipitate | 1.0 – 1.5 |
| Red precipitate | 1.5 – 2.0 |
Important Note
- A positive Benedict’s test does not always mean glucose is present.
- False positive reactions may occur with:
- Vitamin C
- Chloroform
- Formaldehyde
- Lactic acid
- Dextrin
- In pregnant or lactating women, lactose may also cause positive Benedict’s test.
Clinical Interpretation
Physiological Glycosuria
Sugar may appear in urine temporarily even without disease when blood glucose rises briefly above renal threshold.
Causes
- Excess carbohydrate intake
- Emotional stress
- Pregnancy
- After heavy exercise
Pathological Glycosuria
Persistent sugar in urine usually indicates disease.
Causes
- Diabetes Mellitus
- Renal glycosuria
- Endocrine disorders such as hyperthyroidism
- Cushing’s syndrome
Special Sugars Seen in Urine
- Lactose may appear during pregnancy and lactation
- Galactose may appear in galactose metabolism disorders
- Fructose may appear in fructosuria
Protein
A. Sulphosalicylic Acid Test
Principle
- Proteins react with sulphosalicylic acid to form insoluble protein sulphosalicylate causing turbidity.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 2 mL clear urine | — | — |
| Add few drops of 20% sulphosalicylic acid | Turbidity appears | Protein present |
B. Heat Coagulation Test
Principle
- On heating, proteins denature and coagulate.
- Phosphates may also produce turbidity, but dissolve with acetic acid.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Fill test tube 3/4 with clear urine | — | — |
| Heat upper one-third of urine | Turbidity in heated part | Protein or phosphate present |
| Add 1–2 drops of 1% acetic acid | Turbidity persists | Protein confirmed |
C. Heller’s Test
Principle
- Protein reacts with concentrated nitric acid and forms a white ring at the junction of two liquids.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 3 mL concentrated nitric acid in a test tube | — | — |
| Carefully add equal amount of urine along the side of the test tube | White ring at junction | Protein present |
Clinical Interpretation
Physiological Proteinuria
Protein may appear temporarily in urine under normal non-disease conditions.
Causes
- Strenuous exercise
- Pregnancy
- Emotional stress
- Exposure to cold
- High fever
- Standing for long time (postural proteinuria)
Pathological Proteinuria
Persistent protein in urine usually indicates disease.
Renal Causes
- Nephritis
- Nephrosis
- Diabetic nephropathy
- Glomerular damage
Post-Renal Causes
- Urinary tract infection
- Tuberculosis
- Tumors of urinary tract
Systemic Causes
- Hypertension
- Congestive heart failure
Special Protein
- Multiple Myeloma → Bence Jones protein present
Acetone Bodies (Ketone Bodies)
Rothera’s Test
Principle
- Acetone and acetoacetic acid react with sodium nitroprusside in presence of ammonia to form a purple ring.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Saturate 5 mL urine with ammonium sulfate crystals | — | — |
| Add 2 drops fresh 5% sodium nitroprusside | — | — |
| Add 1 mL strong ammonia slowly along the side | Purple ring appears | Acetone or acetoacetic acid present |
Gerhardt’s Test
Principle
- Acetoacetic acid reacts with ferric chloride to produce port-wine color.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 3 mL urine | — | — |
| Add few drops of 10% ferric chloride solution | Port-wine color appears | Acetoacetic acid present |
Important Notes
- If Gerhardt’s test is negative and Rothera’s test is positive, acetone is present.
- Alkaline urine interferes with Gerhardt’s test because ferric hydroxide may form.
- Urine should be neutralized with 1% acetic acid before testing.
- Salicylic acid also gives similar color in Gerhardt’s test.
- Boiling urine removes acetoacetic acid but not salicylic acid.
Clinical Interpretation
Physiological Ketonuria
Small temporary ketone excretion may occur under normal conditions when carbohydrate supply is reduced.
Causes
- Fasting
- Starvation
- Prolonged exercise
- Low carbohydrate diet
- Pregnancy
Pathological Ketonuria
Persistent ketone bodies in urine usually indicate abnormal fat metabolism.
Causes
- Diabetes Mellitus (especially diabetic ketoacidosis)
- Severe vomiting
- Fever
- Severe anemia
- Phosphorus poisoning
Bile Salts
Hay’s Test
Principle
- Bile salts reduce the surface tension of urine, allowing sulfur powder to sink.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Fill 2/3 of one test tube with urine and another with water (control) | — | — |
| Sprinkle sulfur powder on both tubes | Sulfur sinks in urine but floats on water | Bile salts present in urine |
Clinical Interpretation
Physiological
- Bile salts are absent in normal urine.
Pathological
- Present in:
- Obstructive jaundice
- Hepatocellular jaundice
Bile Pigments
Fouchet’s Test
Principle
- Barium chloride reacts with sulfate radicals present in urine to form barium sulfate precipitate.
- If Bilirubin is present, it adheres to the precipitate.
- On adding Fouchet’s reagent, bilirubin is oxidized from yellow bilirubin to green biliverdin.
- This oxidation occurs due to ferric chloride in the presence of trichloroacetic acid.
- Appearance of green or blue-green color indicates bile pigment in urine.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 5 mL urine and add 1–2 drops magnesium sulfate with 5 mL barium chloride | Precipitate forms | — |
| Filter the precipitate | — | — |
| Add Fouchet’s reagent on precipitate | Blue or green color appears | Bile pigment present |
Clinical Interpretation
Physiological
- Bilirubin is absent in normal urine.
Pathological
- Present in:
- Hepatocellular jaundice
- Obstructive jaundice
- Liver disease
Urobilinogen
Ehrlich’s Test
Principle
- Urobilinogen reacts with Ehrlich’s reagent to produce red color.
- The color becomes more intense after addition of sodium acetate.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 2 mL fresh urine in a test tube | — | — |
| Add 2 mL Ehrlich’s reagent and mix | Pink to red color develops | Urobilinogen present |
| Add sodium acetate till saturation | Deep red color if increased | Increased urobilinogen |
Result Interpretation
| Result | Interpretation |
|---|---|
| Red color | Increased urobilinogen |
| Pink color | Normal urobilinogen |
| No difference from control | Urobilinogen absent |
Clinical Interpretation
Physiological
- Small amount normally present in urine.
Pathological
Increased Urobilinogen
- Hemolytic jaundice
- Liver dysfunction
Absent Urobilinogen
- Complete biliary obstruction
Blood Pigments
Benzidine Test
Principle
- Heme of hemoglobin decomposes hydrogen peroxide to release nascent oxygen.
- Nascent oxygen oxidizes benzidine to produce bluish-green color.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Dissolve a pinch of benzidine in 1 mL glacial acetic acid | — | — |
| Add 1 mL hydrogen peroxide | — | — |
| Add 5–10 drops of urine | Deep blue color appears | Blood pigment present |
Control Test
| Procedure | Observation | Interpretation |
|---|---|---|
| Repeat test using water instead of urine | No blue color | Negative control |
Guaiac Test
Principle
- Hemoglobin acts as pseudo-peroxidase and oxidizes guaiac reagent in presence of hydrogen peroxide.
Procedure, Observation and Interpretation
| Procedure | Observation | Interpretation |
|---|---|---|
| Take 5 mL fresh urine | — | — |
| Add gum guaiac reagent till turbidity appears | — | — |
| Add 10 drops hydrogen peroxide | Blue color develops | Blood pigment present |
| Boil urine for 15–20 minutes | Blue remains = positive | Hemoglobin present |
Clinical Interpretation
Hematuria (Intact RBCs in Urine)
Causes
- Injury to urinary tract or kidney
- Urinary tract infection
- Tumors of kidney or urinary tract
- Urinary calculus
- Enlarged prostate
Hemoglobinuria (Free Hemoglobin in Urine)
Causes
- Incompatible blood transfusion
- Hemolytic jaundice
- Severe burns
- Malaria
- Snake bite
- Sickle Cell Disease
- Severe muscular exertion

