Introduction
- Colorimetry and Spectrophotometry are among the most important analytical techniques used in laboratory science.
- These methods are widely used in clinical biochemistry, microbiology, pathology, pharmacology, environmental science, and research laboratories because many chemical substances can be measured accurately by observing how they absorb light.
- In practical laboratory work, many biochemical tests such as glucose estimation, urea estimation, creatinine estimation, bilirubin estimation, enzyme analysis, and protein estimation depend on these instruments.
- Although both instruments work on light absorption principles, their sensitivity, wavelength range, and precision differ significantly.
Principle of Light Absorption
- When light passes through a colored solution:
- Some light is absorbed by the solution
- Some light is transmitted through the solution
- The amount of absorbed light depends on:
- Concentration of colored substance
- Thickness of solution
- Wavelength of incident light
- Darker solution absorbs more light and transmits less light.
Beer-Lambert Law
- Both colorimeter and spectrophotometer work according to Beer-Lambert Law.
Statement
- The absorbance of light by a solution is directly proportional to:
- Concentration of absorbing substance
- Path length of light through the solution
Formula
A = log I₀ / I
Where:
- A = Absorbance
- I₀ = Intensity of incident light
- I = Intensity of transmitted light
Components of Colorimeter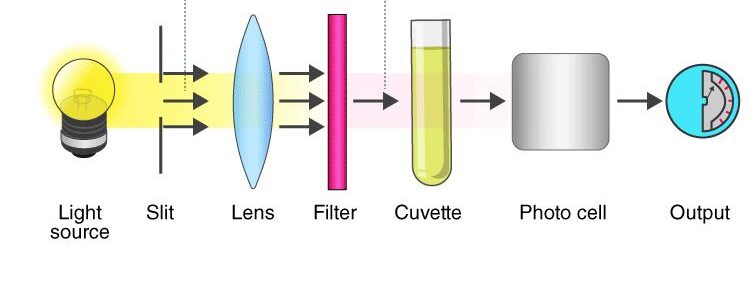
1. Light Source (Radiant Energy)
The light source must provide stable, continuous radiation across the visible spectrum.
-
Tungsten Filament Lamp: This is the most common source. It operates by heating a tungsten wire until it glows, emitting light in the 330 nm to 900 nm range.
-
Halogen Lamps: Often used for increased brightness and a longer lifespan.
-
Importance of Stability: Even a slight fluctuation in voltage can change the light intensity, leading to errors. Modern instruments use voltage regulators to keep the output constant.
2. Slit
- The slit is an important component of a spectrophotometer because it controls the amount and width of light entering the optical system.
- It ensures that only a narrow beam of light passes through the instrument for accurate measurement.
3. Monochromator / Filter (Wavelength Selection)
A colorimeter uses filters to isolate a specific band of wavelengths. This is crucial because a substance absorbs light most efficiently at its “Absorption Maximum”.
-
Absorption Filters: Made of colored glass or gelatin. They work by absorbing unwanted colors and transmitting only the required color. They typically have a wide bandwidth (20–50 nm).
-
Interference Filters: These use silver-coated dielectric layers to reflect unwanted wavelengths, allowing for a much narrower bandwidth (5–10 nm), which significantly increases the instrument’s sensitivity.
Complementary Colors Used in Colorimetry with Wavelength Range
| Color of Solution | Why It Appears This Color | Color Absorbed Most Strongly | Filter Used | Approximate Wavelength (nm) |
|---|---|---|---|---|
| Blue | Transmits blue light | Yellow | Yellow filter | 570–590 nm |
| Red | Transmits red light | Green | Green filter | 495–570 nm |
| Green | Transmits green light | Red | Red filter | 620–750 nm |
| Yellow | Transmits yellow light | Blue | Blue filter | 450–495 nm |
| Orange | Transmits orange light | Blue-green | Blue-green filter | 490–520 nm |
| Violet | Transmits violet light | Yellow-green | Green filter | 560–580 nm |
4. Sample Holder (The Cuvette)
The cuvette is the vessel that holds your sample. Its quality directly impacts the “Optical Density” (OD).
-
Geometry: Most cuvettes are square with a 1 cm internal path length. This standardization allows the use of the molar absorptivity constant in calculations.
-
Optical Surface: Only two sides are transparent; the other two are usually “frosted” to allow for handling without leaving fingerprints on the light path.
-
Materials: * Glass: Standard for visible light.
-
Plastic: Disposable and prevents cross-contamination.
-
5. Photodetector (Conversion System)
The detector senses the light that has passed through the cuvette and converts it into an electrical current.
-
-
Photovoltaic (Barrier-Layer) Cell: These consist of a metal base plate (like iron) coated with a layer of selenium. When photons hit the selenium, electrons are released.
-
Linearity: A good detector must be linear, meaning the electrical output must be directly proportional to the intensity of the light hitting it.
-
6. Readout / Display
The electrical signal from the detector is sent to a galvanometer or a digital processor.
-
The “Blanking” Process: The readout allows the user to set the instrument to zero using a “blank” (usually distilled water or a reagent without the sample). This cancels out any light absorption caused by the cuvette or the solvent itself.
-
Dual Scales: Most displays show both Absorbance (logarithmic scale) and Percentage Transmittance (linear scale).
Working of Colorimeter
1. Preparation and Warm-up
-
Power On: Switch on the instrument and allow it to warm up for about 10 to 15 minutes. This stabilizes the light source and the detector circuit to prevent “drift” during readings.
-
Wavelength Selection: Rotate the filter wheel to select the appropriate wavelength for the specific analyte. This is typically the complementary color of the solution.
2. Standardizing the Instrument (Blanking)
-
Clean the Cuvette: Take a clean cuvette and fill it with a “Blank” solution (usually distilled water or the reagent without the sample).
-
Alignment: Wipe the outer surface of the cuvette with a lint-free tissue to remove fingerprints or moisture. Place it in the cuvette holder, ensuring the clear sides are in the light path.
-
Set Zero: Adjust the control knob (or press the “Auto-Zero” button) so that the display reads 0.00 Absorbance or 100% Transmittance. This step subtracts any light absorbed by the solvent or the glass.
3. Measuring the Standard
-
Rinse and Fill: Remove the blank, rinse the cuvette with a small amount of the standard solution (a solution of known concentration), and then fill it.
-
Read and Record: Place the standard in the holder and record the absorbance value. This value is used as a reference point for your calculations.
4. Measuring the Unknown Sample
-
Sample Loading: Rinse the cuvette again and fill it with the unknown sample solution.
-
Record Absorbance: Place the sample in the instrument and record the absorbance.
-
Verification: If testing multiple samples, it is good practice to re-check the “Blank” periodically to ensure the instrument hasn’t drifted.
5. Calculation of Concentration
The concentration of the unknown sample is calculated using the ratio of the absorbances, based on the Beer-Lambert Law
Ct = At / As x Cs
Where:
-
Ct: Concentration of the unknown sample.
-
At: Absorbance of the unknown sample.
-
As: Absorbance of the standard.
-
Cs: Concentration of the standard.
6. Shutdown
-
Cleaning: Remove the cuvette, discard the solution properly, and wash the cuvette with distilled water.
-
Storage: Turn off the power and cover the instrument to protect the optical components from dust.
Uses of Colorimeter
In clinical and industrial settings, colorimeters are used whenever a chemical reaction produces a colored compound.
1. Clinical Biochemistry (Most Common)
-
Blood Chemistry: Measuring concentrations of glucose, urea, creatinine, bilirubin, and cholesterol in serum or plasma.
-
Proteins: Estimating total protein or albumin levels using color-changing reagents (like the Biuret test).
-
Enzymes: Monitoring liver or heart enzymes by measuring the rate of color change over time.
2. Hematology
-
Hemoglobin Estimation: Measuring hemoglobin levels (cyanmethemoglobin method) to diagnose anemia.
3. Industrial and Environmental Uses
-
Water Quality: Testing for levels of chlorine, fluoride, or heavy metals in drinking water.
-
Food Industry: Standardizing the color of juices, jams, and wine to ensure consistency.
-
Textiles: Monitoring dye concentrations in fabrics.
Advantages of Colorimeter
-
Ease of Use: It is one of the simplest optical instruments to operate, making it ideal for undergraduate medical labs and small clinics.
-
Cost-Effective: Compared to a Spectrophotometer or an HPLC, colorimeters are very inexpensive to purchase and maintain.
-
Durability: With fewer moving parts and a robust design, they can withstand heavy daily use in busy environments.
-
Speed: Once calibrated, a reading can be taken in seconds, allowing for high-throughput testing in routine labs.
-
Portability: Many modern colorimeters are battery-operated and handheld, allowing for field testing (like checking water quality at a remote site).
Limitations of Colorimeter
-
Visible Light Only: Colorimeters can only measure substances that absorb light in the visible spectrum (380–750 nm). They cannot be used for substances that only absorb UV light (like DNA or certain drugs).
-
Broad Bandwidth: Because they use filters rather than a diffraction grating, they have a wider “spectral bandwidth.” This means they are less precise than spectrophotometers when measuring complex mixtures.
-
Limited Sensitivity: They are not suitable for measuring substances present in extremely low (trace) concentrations.
-
Requires Color: The analyte must naturally have a color or be capable of reacting with a reagent to produce a color. If no color change occurs, the colorimeter is useless.
-
Interference: If a sample is “turbid” (cloudy or has particles), the particles will scatter light, leading to a falsely high absorbance reading.
Components of a Spectrophotometer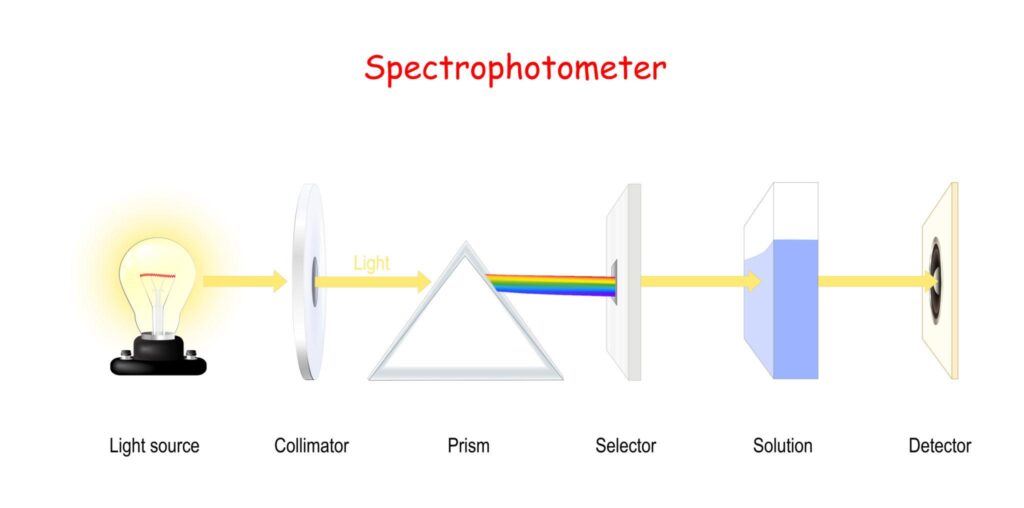
1. Light Source (Radiant Energy Source)
A spectrophotometer often uses two different lamps to cover a wide range of wavelengths:
-
Tungsten Lamp: Provides light for the visible spectrum (380–750 nm).
-
Deuterium Lamp: Provides light for the ultraviolet (UV) spectrum (190–380 nm).
-
Function: It must provide stable, high-intensity radiation across the desired range.
2. Slit
- The slit is an important component of a spectrophotometer because it controls the amount and width of light entering the optical system.
- It ensures that only a narrow beam of light passes through the instrument for accurate measurement.
3. Monochromator
This is the “heart” of the instrument. Unlike a colorimeter’s filter, a monochromator can “tune” to any specific wavelength.
-
Entrance Slit: Narrows the light into a thin beam.
-
Dispersing Element (Prism or Diffraction Grating): This component breaks the white light into its component colors (the rainbow). A diffraction grating is most common in modern units because it is highly precise.
-
Exit Slit: A moveable slit that allows only a very specific, narrow wavelength (e.g., exactly 546 nm) to pass through to the sample.
4. Sample Holder (Cuvette)
Since spectrophotometers often work in the UV range, the material of the cuvette is critical:
-
Glass/Plastic: Used only for visible light.
-
Quartz/Fused Silica: Must be used for UV light because standard glass absorbs UV radiation and would interfere with the reading.
-
Path Length: Usually standardized at 1 cm.
5. Detector (Photometric System)
Because the light intensity can be very low at specific narrow wavelengths, more sensitive detectors are used than those in basic colorimeters.
-
Photomultiplier Tube (PMT): The most common detector. It not only detects photons but multiplies the signal, making it sensitive enough to detect even trace amounts of a substance.
-
Photodiode Array (PDA): Used in advanced units to measure all wavelengths simultaneously.
6. Computing and Readout Device
The electrical signal from the detector is processed by a microprocessor.
-
Function: It converts the signal into Absorbance (A), Transmittance (%T), or direct Concentration.
-
Display: Digital screens often show a “scanning” graph (a spectrum) showing absorbance across a range of wavelengths rather than just a single number.
Uses of Spectrophotometer
- Quantitative estimation of biochemical substances such as glucose, urea, creatinine, bilirubin, and cholesterol
- Measurement of enzyme activity in clinical biochemistry
- Estimation of proteins in serum, urine, and cerebrospinal fluid
- Determination of DNA and RNA concentration in molecular studies
- Drug analysis in pharmaceutical laboratories
- Assessment of bacterial growth in microbiology by turbidity measurement
- Water quality and environmental chemical analysis
- Routine use in research laboratories for analytical investigations
Advantages of Spectrophotometer
1. Wide Spectral Range
Unlike a colorimeter, which is limited to visible light (400–700 nm), a spectrophotometer can operate in the Ultraviolet (UV) range (190–400nm).
-
Clinical Significance: This allows for the measurement of substances that do not have a visible color, such as DNA, RNA, and many drugs, which absorb light in the UV region.
2. High Wavelength Precision
Because it uses a diffraction grating or prism instead of fixed filters, you can select any specific wavelength for NADH-based enzyme assays.
-
Narrow Bandwidth: It provides a very “pure” light beam (1–2 nm wide), which reduces interference from other substances in the sample.
3. High Sensitivity
Spectrophotometers typically use a Photomultiplier Tube (PMT) as a detector.
-
This component is extremely sensitive and can detect very low intensities of light. This allows medical professionals to measure substances present in minute concentrations (trace elements or hormones) that a colorimeter would miss.
4. Quantitative and Qualitative Analysis
-
Quantitative: It accurately measures “how much” of a substance is present.
-
Qualitative (Scanning): It can perform a spectral scan, measuring absorbance across a range of wavelengths to create a “fingerprint” of a substance. This helps identify unknown compounds or check the purity of a sample.
5. Reduced Human Error (Automation)
Modern spectrophotometers are highly automated. They can:
-
Automatically perform “blanking” and calibration.
-
Store standard curves in memory so you don’t have to run a standard every single time.
-
Use a “sipper system” or “micro-cuvettes” to measure very small volumes of expensive reagents.
6. Versatility in Sample Types
It can accommodate various types of cuvettes, including:
-
Micro-cuvettes: For samples as small as 1 mu L (useful in pediatrics or forensic science).
-
Flow cells: For continuous monitoring of reactions.
Difference Between Colorimeter and Spectrophotometer
| Feature | Colorimeter | Spectrophotometer |
|---|---|---|
| Definition | Instrument used to measure concentration of colored solutions in visible range | Advanced instrument used to measure absorbance over a wide wavelength range |
| Principle | Measures absorption of visible light by colored solution | Measures absorption of monochromatic light in UV, visible, and sometimes IR range |
| Wavelength Range | Visible light only (400–700 nm) | UV + Visible + IR range |
| Light Source | Tungsten lamp | Tungsten lamp and deuterium lamp |
| Wavelength Selection | Uses colored filters | Uses monochromator (prism or diffraction grating) |
| Sensitivity | Less sensitive | Highly sensitive |
| Accuracy | Moderate accuracy | High accuracy |
| Cuvette Used | Glass cuvette | Glass or quartz cuvette |
| Cost | Less expensive | More expensive |
| Applications | Routine biochemical tests | Advanced biochemical and molecular analysis |