Introduction
- Proteins are one of the most important macronutrients required for normal growth, development, and maintenance of the human body.
- They provide amino acids, which are the basic building blocks needed for synthesis of body proteins.
- Every living cell depends on proteins for structural stability and metabolic activity.
- Structural proteins help maintain tissue architecture, while functional proteins regulate biochemical reactions inside cells.
- Because proteins are large complex macromolecules, they cannot be absorbed directly through the intestinal mucosa.
- Therefore, dietary proteins must first undergo digestion to convert them into smaller molecules.
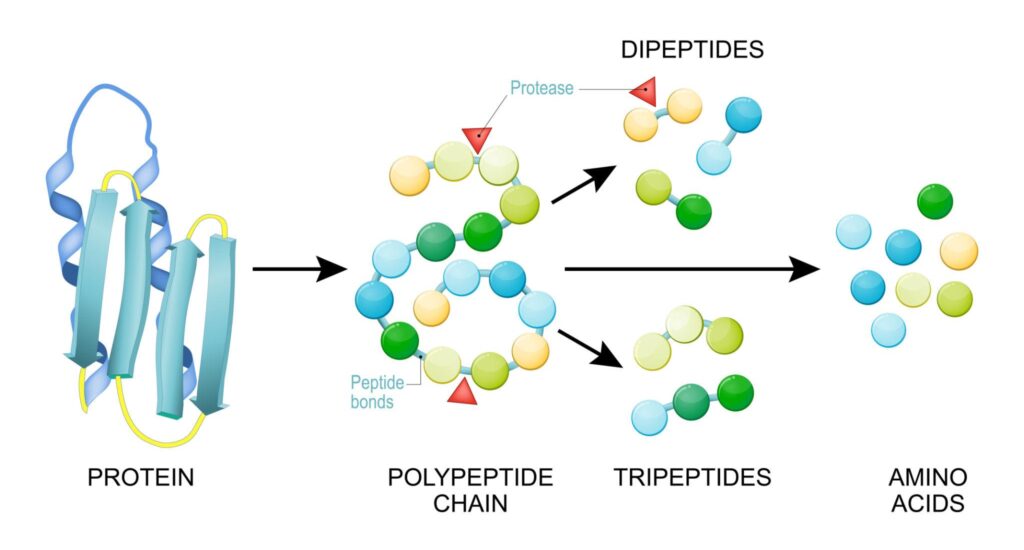
- During digestion, proteins are gradually broken down into:
- Large polypeptides
- Small peptides
- Dipeptides
- Tripeptides
- Free amino acids
- Only amino acids and very small peptides can be absorbed through intestinal epithelial cells.
- Protein digestion is therefore essential for proper nutritional utilization of dietary proteins.
- Protein digestion is a highly coordinated biochemical process involving several digestive organs and enzymes.
- It requires:
- Hydrochloric acid in the stomach
- Gastric proteolytic enzymes
- Pancreatic proteolytic enzymes
- Brush border peptidases of intestinal mucosa
- Specialized transport systems in enterocytes
- Each digestive enzyme acts at a specific site and pH to ensure complete hydrolysis of peptide bonds.
- The final products of digestion are absorbed mainly in the small intestine and transported to the liver through portal circulation.
- Efficient digestion and absorption of protein are essential for:
- Maintaining nitrogen balance
- Protein synthesis
- Energy production when required
- Formation of important biological compounds
- Any defect in protein digestion or absorption may lead to malnutrition, hypoproteinemia, and metabolic disturbances.
Dietary Proteins: Nature, Composition and Nutritional Importance
Composition of Dietary Proteins
- Proteins are composed of 20 standard amino acids.
- Amino acids are linked by peptide bonds formed between amino and carboxyl groups.
- The sequence of amino acids determines protein structure and biological function.
Nutritional Classification of Proteins
Complete proteins
Contain all essential amino acids in adequate quantity.
Examples
- Milk proteins
- Egg proteins
- Soy protein
Incomplete proteins
Deficient in one or more essential amino acids.
Examples
- Gelatin
- Some cereal proteins
Essential Amino Acids
These cannot be synthesized by the body and must be supplied through diet.
Importance
- Required for growth
- Tissue repair
- Enzyme synthesis
- Neurotransmitter production
Phases of Protein Digestion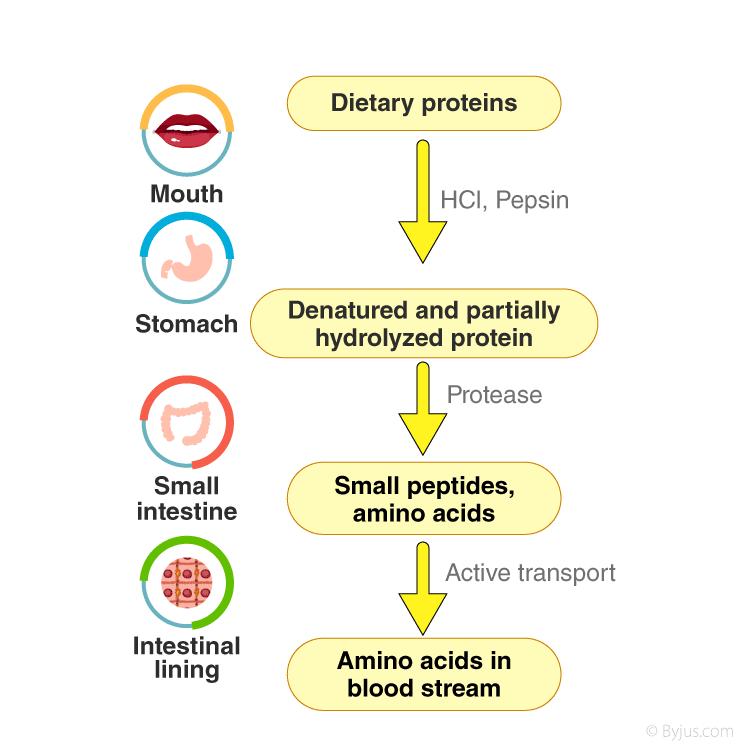
| Phase | Site of Action | Major Enzymes | Optimum pH | End Products |
|---|---|---|---|---|
| Gastric Phase | Stomach | Pepsin | 1.5–2.5 | Polypeptides, Peptones |
| Pancreatic Phase | Duodenum | Trypsin, Chymotrypsin, Elastase, Carboxypeptidase | 7.5–8.5 | Oligopeptides |
| Intestinal Phase | Jejunum, Ileum | Aminopeptidase, Dipeptidase, Tripeptidase | 7.5–8.0 | Free amino acids, small peptides |
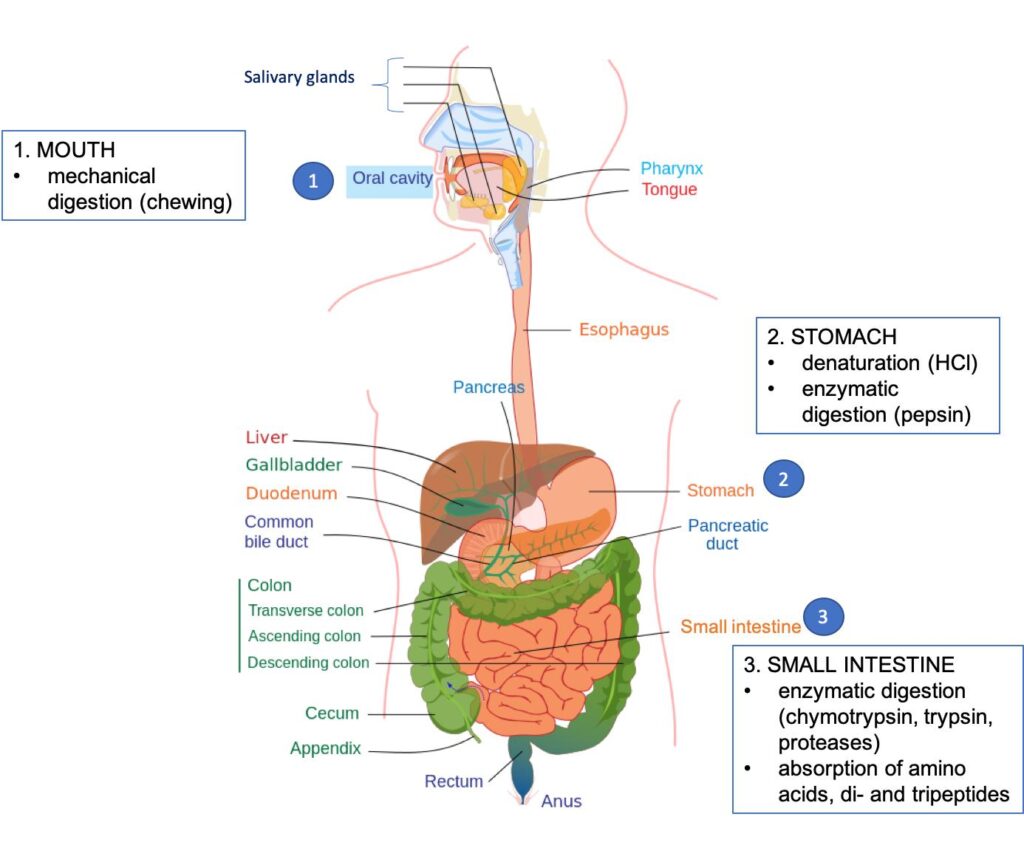
Digestion of Protein in Mouth
- No significant chemical digestion of protein occurs in the mouth because saliva does not contain any proteolytic enzyme capable of hydrolyzing peptide bonds.
- The mouth mainly performs mechanical digestion, which is the first step in preparing proteins for later enzymatic digestion in the stomach and intestine.
Role of Mastication
- Mastication (chewing) breaks food into smaller particles.
- This increases the surface area of food exposed to digestive enzymes later.
- Proper chewing helps mix food uniformly with saliva and forms a soft bolus suitable for swallowing.
Role of Saliva
- Saliva moistens and lubricates food.
- It helps in bolus formation and smooth passage through the esophagus.
- Saliva contains:
- Water
- Mucus
- Electrolytes
- Salivary amylase
- However, salivary amylase acts only on carbohydrates, not on proteins.
Why Protein Digestion Does Not Start in Mouth
- Proteolytic enzymes require an acidic medium for activation.
- The oral cavity has a neutral pH, which is unsuitable for gastric proteases like pepsin.
- Therefore protein digestion begins only after food reaches the stomach.
Importance of Oral Phase in Protein Digestion
- Although chemical digestion does not occur, the oral phase is important because:
- It improves later gastric digestion
- Facilitates effective mixing with gastric acid and enzymes
- Helps proper swallowing and gastric processing
- Thus, the mouth contributes mainly by mechanical preparation of dietary proteins for subsequent digestion.
Digestion of Protein in Stomach
Gastric Phase
- The stomach is the first major site of chemical digestion of proteins.
- In the stomach, dietary proteins are exposed to hydrochloric acid (HCl) and proteolytic enzymes, which initiate protein breakdown.
- Gastric digestion converts large protein molecules into proteoses, peptones, and polypeptides.
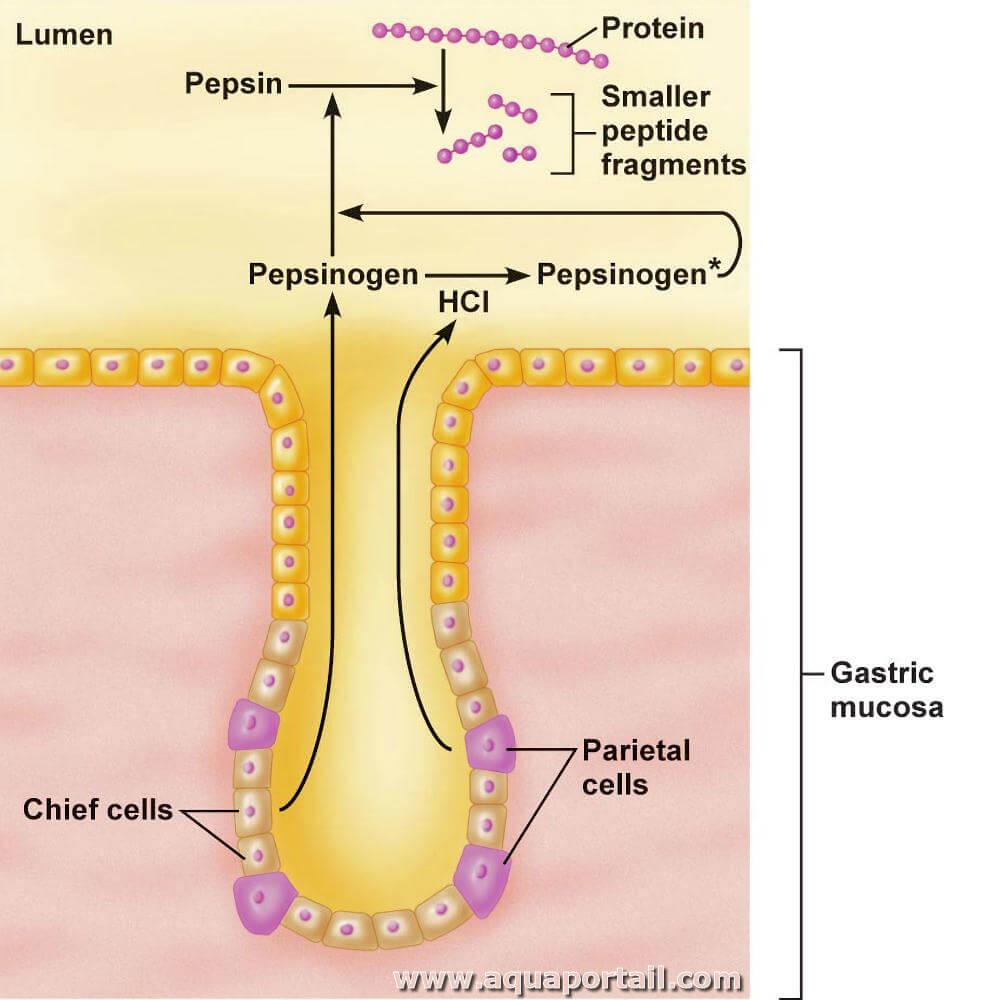
Role of Hydrochloric Acid (HCl)
- Hydrochloric acid is secreted by parietal cells of gastric glands.
- It produces a highly acidic environment with pH around 1.5–2.0, which is essential for protein digestion.
Functions of HCl
- Denatures proteins by unfolding their secondary and tertiary structure
- Converts pepsinogen into pepsin
- Provides optimum acidic pH for pepsin activity
- Kills many microorganisms present in food
- Softens connective tissue present in dietary proteins
Denaturation of Proteins
- Dietary proteins normally have compact folded structure.
- In acidic medium, this structure is disrupted.
Result
- Internal peptide bonds become exposed
- Enzymes can attack proteins more easily
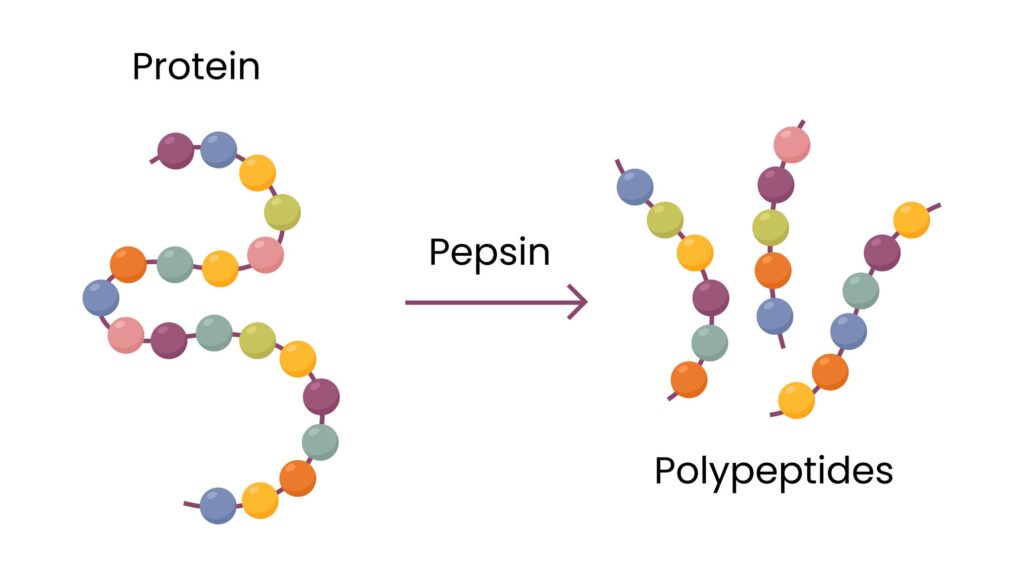
Pepsin: Major Gastric Proteolytic Enzyme
- Pepsin is the main enzyme responsible for gastric protein digestion.
- It is secreted as inactive pepsinogen by chief cells.
Activation of Pepsinogen
Pepsinogen + HCl → Pepsin
- Hydrochloric acid removes inhibitory peptide from pepsinogen and converts it into active pepsin.
- Activated pepsin also activates additional pepsinogen molecules (autocatalysis).
Nature of Pepsin
- Pepsin is an endopeptidase
- It acts on internal peptide bonds within protein molecules
Bonds Hydrolyzed by Pepsin
Pepsin preferentially breaks peptide bonds involving:
- Phenylalanine
- Tyrosine
- Tryptophan
These are aromatic amino acids.
Products Formed by Pepsin
- Proteoses
- Peptones
- Large polypeptides
Pepsin does not produce free amino acids.
Optimum pH of Pepsin
- Maximum activity occurs at pH 1.5–2.0
- Pepsin becomes inactive when pH rises above 5
Gastric Mixing and Churning
- Stomach muscles continuously mix food with gastric juice.
- This mechanical movement increases contact between proteins and digestive enzymes.
Result
- Semi-liquid mixture called chyme is formed.
Rennin (Chymosin) in Infants
- In infants, rennin (chymosin) helps digest milk protein.
Function of Rennin
- Converts soluble casein into insoluble calcium paracaseinate
Digestion of Protein in Small Intestine
- The small intestine is the principal site of protein digestion, where most dietary proteins are converted into absorbable amino acids and small peptides.
- Protein digestion in the small intestine begins when acidic chyme from the stomach enters the duodenum and mixes with pancreatic juice, bile, and intestinal secretions.
- Pancreatic enzymes perform the major part of protein hydrolysis because gastric digestion in the stomach remains incomplete.
Neutralization of Gastric Acid
- The acidic chyme entering the duodenum must first be neutralized because pancreatic enzymes work best in alkaline medium.
- Bicarbonate ions (HCO₃⁻) present in pancreatic juice neutralize hydrochloric acid.
- Raises intestinal pH to about 7.5–8.0
- Provides optimum pH for pancreatic proteases
- Protects intestinal mucosa from acid injury
Pancreatic Phase
Pancreatic Proteolytic Enzymes
- Pancreas secretes proteolytic enzymes in inactive precursor form (zymogens) to prevent autodigestion of pancreatic tissue.
Main Pancreatic Zymogens
- Trypsinogen
- Chymotrypsinogen
- Proelastase
- Procarboxypeptidase A
- Procarboxypeptidase B
Why Secreted as Inactive Form
- Active proteases inside pancreas would digest pancreatic proteins.
- Therefore activation occurs only after enzymes reach intestine.
Activation of Trypsinogen
- The first step in pancreatic enzyme activation is conversion of trypsinogen into trypsin.
- This occurs by action of enteropeptidase (enterokinase) present on intestinal brush border.
Trypsinogen → Trypsin
Importance of Trypsin
- Trypsin is the central enzyme because it activates all other pancreatic proteases.
Activation Cascade
- Chymotrypsinogen → Chymotrypsin
- Proelastase → Elastase
- Procarboxypeptidase → Carboxypeptidase
Trypsin
- Trypsin is an endopeptidase.
- It hydrolyzes internal peptide bonds.
Specificity of Trypsin
It cleaves peptide bonds after:
- Lysine
- Arginine
These amino acids are basic amino acids.
Products Formed
- Smaller polypeptides
- Oligopeptides
Chymotrypsin
- Chymotrypsin is also an endopeptidase.
Specificity of Chymotrypsin
It acts on peptide bonds containing aromatic amino acids:
- Phenylalanine
- Tyrosine
- Tryptophan
Products Formed
- Smaller peptides
Elastase
- Elastase digests elastin and peptide bonds involving small neutral amino acids.
Amino acids commonly attacked
- Glycine
- Alanine
- Serine
Importance
- Helps digest connective tissue proteins.
Carboxypeptidase
- Carboxypeptidase is an exopeptidase.
Action
- Removes amino acids one by one from carboxyl terminal end of peptide chain.
Types
- Carboxypeptidase A
- Carboxypeptidase B
Result
- Produces free amino acids and smaller peptides.
Pancreatic enzyme
| Zymogen (Inactive form) | Active Enzyme | Activator | Major Substrate Bonds Cleaved |
|---|---|---|---|
| Trypsinogen | Trypsin | Enteropeptidase | Lysine, Arginine |
| Chymotrypsinogen | Chymotrypsin | Trypsin | Aromatic AAs (Phe, Tyr, Trp) |
| Proelastase | Elastase | Trypsin | Small neutral AAs (Ala, Gly, Val) |
| Procarboxypeptidase A/B | Carboxypeptidase A/B | Trypsin | C-terminal residues (A or basic AAs) |
Intestinal Phase
The final digestion occurs at the brush border membrane of enterocytes (intestinal epithelial cells).
Major Brush Border Peptidases
| Enzyme | Action | Products Formed |
|---|---|---|
| Aminopeptidase | Removes amino acids from N-terminus | Shorter peptides, amino acids |
| Dipeptidase / Tripeptidase | Splits di- and tripeptides | Free amino acids |
| Enteropeptidase | Converts trypsinogen → trypsin | Initiates activation cascade |
These enzymes complete the hydrolysis, producing free amino acids, dipeptides, and tripeptides suitable for absorption.
Absorption of Protein
- Absorption of protein means transfer of the final products of protein digestion from the intestinal lumen into intestinal epithelial cells and then into the blood.
- Protein absorption occurs mainly in the small intestine, especially in the jejunum and ileum, where intestinal villi and microvilli provide a very large absorptive surface area.
- Only small molecules formed after digestion can be absorbed efficiently.
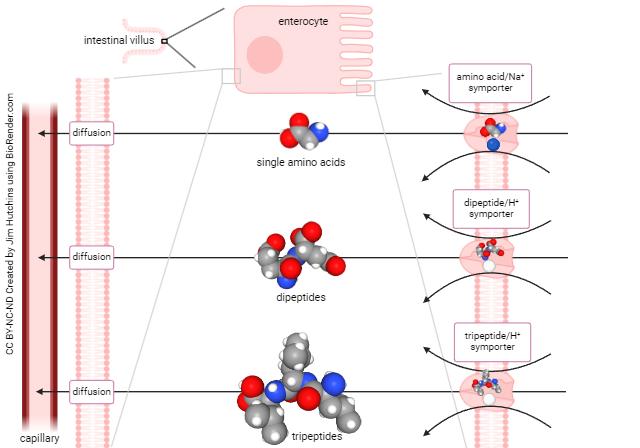
Amino Acid Transport Systems
| System | Main Amino Acids Transported | Mechanism | Clinical Significance |
|---|---|---|---|
| System A | Alanine, Serine, Glycine | Na⁺-dependent active transport | Defect → Hartnup disease |
| System X⁻AG | Aspartate, Glutamate | Na⁺-dependent cotransport | Defect causes acidic aminoaciduria |
| System L | Leucine, Isoleucine, Phenylalanine, Tyrosine | Facilitated diffusion | Important for tissue amino acid exchange |
| System y⁺ | Lysine, Arginine, Ornithine | Na⁺-independent carrier | Defect → Cystinuria |
| System IMINO | Proline, Hydroxyproline | Na⁺-linked transport | Defect causes iminoglycinuria |
Site of Absorption
- Maximum absorption occurs in the jejunum.
- Remaining absorption continues in the ileum.
- The brush border membrane of enterocytes contains specialized transport proteins.
Mechanism of Amino Acid Absorption
- Most amino acids are absorbed by secondary active transport.
Sodium-dependent Cotransport
- Amino acids enter enterocytes together with sodium ions (Na⁺) through specific carrier proteins.
Mechanism
- Sodium concentration inside enterocyte remains low because of Na⁺/K⁺ ATPase pump present on basolateral membrane.
- This sodium gradient provides energy for amino acid entry.
Importance
- Amino acid transport occurs even against concentration gradient.
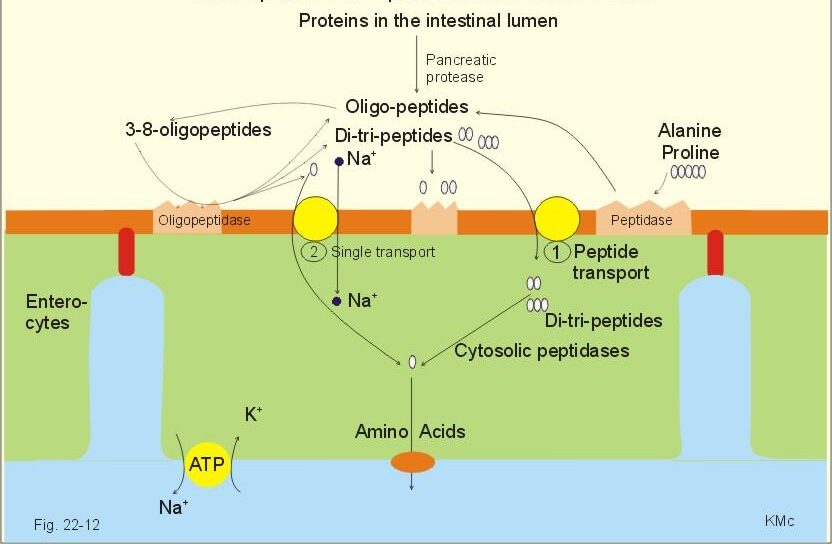
Types of Amino Acid Transport Systems
Different carrier systems exist for different amino acid groups.
Transport systems for
- Neutral amino acids
- Basic amino acids
- Acidic amino acids
- Imino acids
Significance
- Each transporter recognizes specific amino acid groups.
- This ensures efficient absorption of all amino acids.
Absorption of Dipeptides and Tripeptides
- Dipeptides and tripeptides are absorbed more rapidly than free amino acids in many cases.
Mechanism
- They enter enterocytes through H⁺-dependent peptide transporter (PepT1).
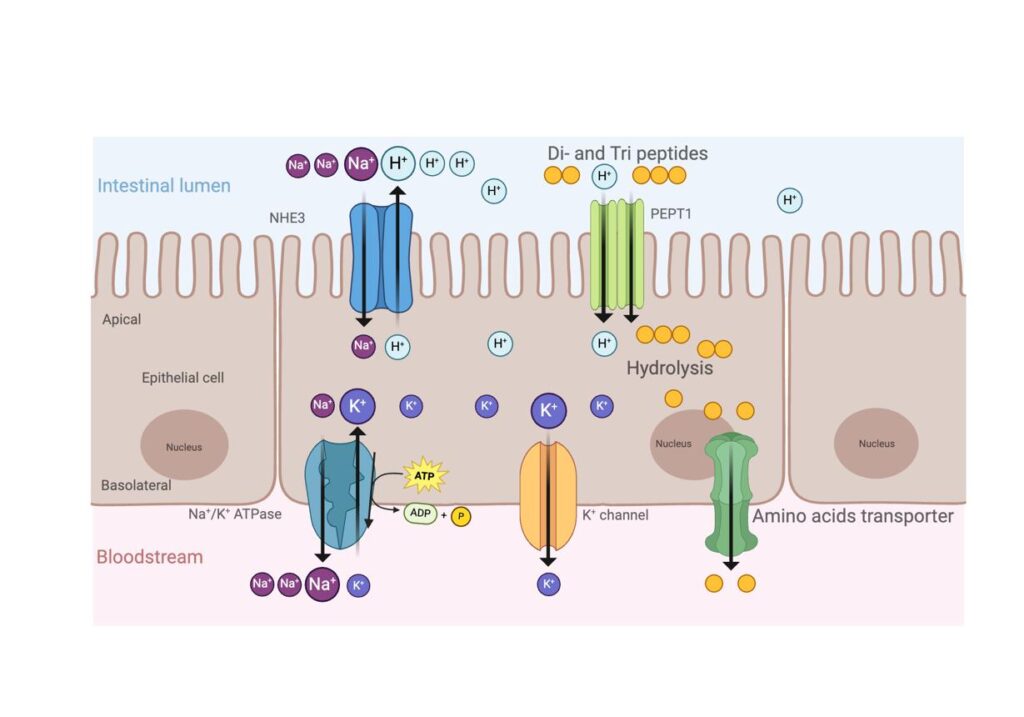
Inside Enterocyte
- Cytoplasmic peptidases hydrolyze peptides into free amino acids.
Result
- Most absorbed protein leaves enterocyte as amino acids.
Transport from Enterocyte to Blood
- Amino acids exit enterocytes through the basolateral membrane by facilitated transport.
Pathway
- Enter interstitial fluid
- Enter capillaries of intestinal villi
- Reach portal vein
- Transported to liver
Fate of Absorbed Amino Acids
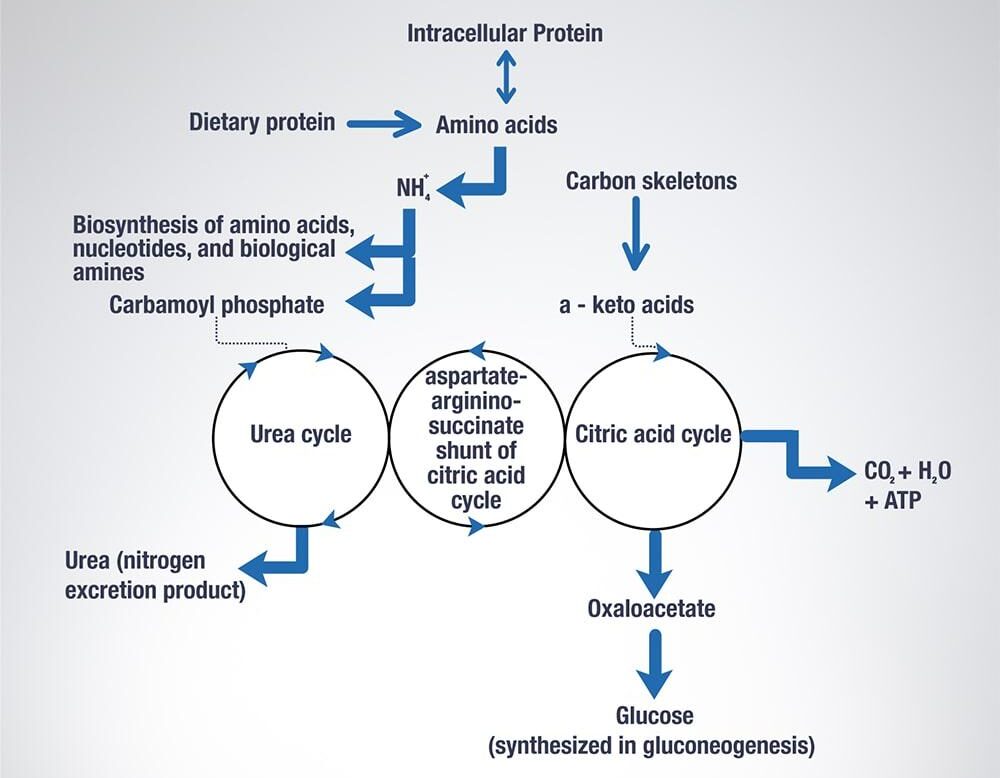
Major uses
- Protein synthesis
- Enzyme formation
- Hormone synthesis
- Plasma protein formation
- Neurotransmitter production
- Formation of nucleotides
Clinical Disorders of Protein Absorption
- Clinical disorders of protein absorption occur when protein digestion is incomplete or when amino acid transport through the intestinal mucosa is defective.
- These disorders reduce the availability of amino acids for normal body functions and may lead to malnutrition, hypoproteinemia, growth retardation, and metabolic abnormalities.
- The defect may occur at different levels:
- Deficiency of digestive enzymes
- Damage to intestinal mucosa
- Defect in amino acid transport systems
1. Pancreatic Insufficiency
- In pancreatic insufficiency, secretion of pancreatic proteolytic enzymes decreases.
- As a result, proteins are not adequately digested in the small intestine.
Causes
- Chronic pancreatitis
- Cystic fibrosis
- Pancreatic duct obstruction
- Pancreatectomy
Effect
- Deficiency of:
- Trypsin
- Chymotrypsin
- Carboxypeptidase
Clinical Features
- Protein maldigestion
- Weight loss
- Muscle wasting
- Malnutrition
2. Enteropeptidase Deficiency
- Enteropeptidase is required for activation of trypsinogen.
Defect
- Trypsinogen cannot convert into trypsin.
Trypsinogen → Trypsin
Result
- All pancreatic protease activation decreases.
Clinical Features
- Severe protein malabsorption
- Diarrhea
- Failure to thrive in infants
3. Celiac Disease
Celiac Disease
- Celiac disease is caused by hypersensitivity to gluten.
Mechanism
- Gluten damages intestinal villi.
- Villous atrophy reduces absorptive surface area.
Result
- Amino acid absorption decreases.
Clinical Features
- Chronic diarrhea
- Weight loss
- Abdominal distension
- Hypoproteinemia
4. Hartnup Disease
- Hartnup disease is an inherited defect of neutral amino acid transport in intestine and kidney.
Amino acids affected
- Tryptophan
- Alanine
- Serine
- Valine
Result
- Reduced intestinal absorption
- Increased urinary loss
Clinical Features
- Pellagra-like dermatitis
- Ataxia
- Neurological symptoms
Reason
- Tryptophan deficiency reduces niacin synthesis.
5. Cystinuria
- Cystinuria is an inherited defect of transport of dibasic amino acids.
Amino acids affected
- Cystine
- Lysine
- Arginine
- Ornithine
Result
- Poor intestinal absorption
- Increased urinary excretion
Clinical Importance
- Cystine is poorly soluble.
- Leads to cystine stone formation in urinary tract.
6. Severe Intestinal Mucosal Damage
Causes
- Severe gastroenteritis
- Inflammatory bowel disease
- Radiation injury
Result
- Amino acid absorption decreases significantly.
7. Protein-Energy Malnutrition
- Prolonged poor protein absorption may lead to severe nutritional deficiency.
Examples
- Kwashiorkor
- Marasmus
Clinical Features
- Growth failure
- Muscle wasting
- Edema
- Hypoproteinemia
| Disorder | Main Defect | Major Effect |
|---|---|---|
| Pancreatic insufficiency | Enzyme deficiency | Protein maldigestion |
| Enteropeptidase deficiency | Trypsin activation defect | Severe malabsorption |
| Celiac disease | Villous atrophy | Reduced amino acid absorption |
| Hartnup disease | Neutral amino acid transport defect | Tryptophan deficiency |
| Cystinuria | Dibasic amino acid transport defect | Cystine stones |