Introduction
- Amino acids are the basic nitrogen-containing units of proteins and are essential for many metabolic processes.
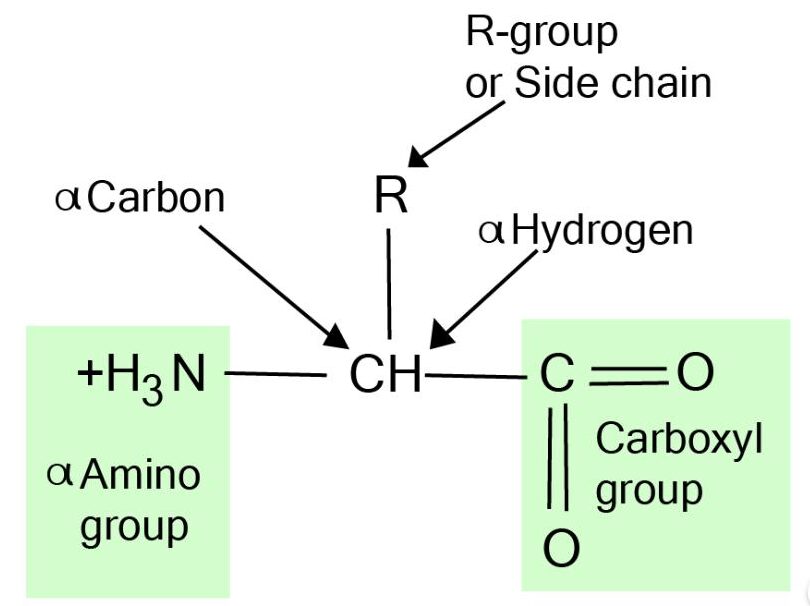
- They participate in important biochemical reactions such as:
- Transamination
- Deamination
- Decarboxylation
- During amino acid breakdown, the amino group is removed and forms ammonia, which is highly toxic to the body.
- Ammonia must be rapidly detoxified to prevent harmful effects, especially on the brain.
- The liver plays a central role in converting ammonia into urea through the urea cycle.
- Urea is a non-toxic compound that is excreted by the kidneys.
- Proper amino acid metabolism is essential for maintaining nitrogen balance and normal body function.
General Reactions of Amino Acids
Transamination
Transamination is the transfer of an amino group (-NH₂) from an amino acid to an α-keto acid, resulting in the formation of a new amino acid and a new α-keto acid.
Amino acid + α-ketoglutarate ⇌ α-keto acid + glutamate
- It is the first step in the metabolism of most amino acids.
- The reaction is catalyzed by aminotransferases (transaminases).
- Pyridoxal phosphate (PLP), derived from vitamin B6, acts as the coenzyme.
- The amino group is usually transferred to α-ketoglutarate, producing glutamate.
Salient Features of Transamination
- Transamination is reversible and occurs in both directions depending on metabolic need.
- No free ammonia is released during the reaction.
- Most amino acids participate, except:
- Lysine
- Threonine
- Proline
- Hydroxyproline
- Occurs in cytoplasm and mitochondria of many tissues.
- Liver is the major site, but reactions also occur in muscle, kidney, and heart.
- Two clinically important enzymes are:
- Alanine aminotransferase (ALT / SGPT)
- Aspartate aminotransferase (AST / SGOT)
- These enzymes are important diagnostic markers in liver disease.
- Transamination helps in:
- Amino acid synthesis
- Amino acid degradation
- Nitrogen transport
Mechanism of Transamination
Transamination occurs in two stages and follows a ping-pong mechanism.
Step 1: Transfer of amino group to PLP
- Amino acid donates its amino group to pyridoxal phosphate (PLP).
- PLP is converted into pyridoxamine phosphate (PMP).
- α-keto acid corresponding to original amino acid is formed.
Amino acid + PLP → Keto acid + PMP
Step 2: Transfer of amino group from PMP
- PMP transfers the amino group to another α-keto acid.
- A new amino acid is formed.
- PLP is regenerated.
PMP + Keto acid → New amino acid + PLP
Example
Alanine + α-ketoglutarate ⇌ Pyruvate + Glutamate
Deamination
Deamination is the removal of the amino group (-NH₂) from an amino acid, resulting in the formation of an α-keto acid and release of ammonia (NH₃).
- It is an important reaction in amino acid catabolism.
- The released ammonia is later converted into urea in the liver.
- Deamination mainly occurs in the liver and kidney.
I. Oxidative Deamination
Oxidative deamination is the removal of an amino group with simultaneous oxidation, producing ammonia and an α-keto acid.
Glutamate + NAD+ + H2O → α-ketoglutarate + NH3 + NADH + H+
Important features
- Mainly occurs in mitochondria of liver and kidney.
- Glutamate is the principal amino acid undergoing oxidative deamination.
- Catalyzed by glutamate dehydrogenase.
- Requires coenzymes:
- NAD⁺ or
- NADP⁺
- It is a reversible reaction.
- Produces free ammonia for urea synthesis.
Importance
- Major source of ammonia in the body
- Connects amino acid metabolism with TCA cycle through α-ketoglutarate
II. Non-Oxidative Deamination
Non-oxidative deamination is removal of the amino group without oxidation and without involvement of oxygen.
Important features
- Occurs mainly with specific amino acids.
- Catalyzed by dehydratases or lyases.
- Requires pyridoxal phosphate (PLP) as coenzyme.
Examples
Serine
Serine → Pyruvate + NH3
Threonine
Threonine → α-ketobutyrate + NH3
Histidine
Histidine → Urocanate + NH3
Importance
- Helps in degradation of certain amino acids
- Produces ammonia directly
Transdeamination
Transdeamination is a combination of:
- Transamination
- Oxidative deamination
This is the major pathway for removal of amino nitrogen from most amino acids.
Sequence:
- Amino acid transfers amino group to α-ketoglutarate
- Glutamate formed
- Glutamate undergoes oxidative deamination
Importance:
- Central mechanism for nitrogen removal
Decarboxylation
Decarboxylation removes the carboxyl group as carbon dioxide.
Amino acid → Amine + CO2
Enzyme:
- Amino acid decarboxylases
Cofactor:
- Pyridoxal phosphate (PLP)
Examples:
- Histidine → Histamine
- Glutamate → GABA
- DOPA → Dopamine
- 5-Hydroxytryptophan → Serotonin
Importance:
- Formation of biologically active amines
Metabolism of Ammonia
- Ammonia (NH₃) is produced during amino acid metabolism.
- It is highly toxic, especially to the nervous system.
- Therefore, ammonia must be rapidly transported and detoxified.
- It is mainly carried as glutamine and alanine.
- Liver converts ammonia into urea, which is excreted by kidneys.
- Liver, kidney, intestine, and muscle are important organs in ammonia metabolism.
Formation of Ammonia
Ammonia is generated in different tissues through several metabolic reactions.
1. Oxidative Deamination
The most important source of ammonia is oxidative deamination of glutamate.
Glutamate + NAD+ + H2O → α-ketoglutarate + NH3 + NADH + H+
- Catalyzed by glutamate dehydrogenase
- Occurs mainly in mitochondria of liver and kidney
- Produces free ammonia directly
- α-ketoglutarate enters TCA cycle
2. Non-Oxidative Deamination
Certain amino acids release ammonia directly without oxidation.
Examples:
- Serine → Pyruvate + NH₃
- Threonine → α-ketobutyrate + NH₃
- Catalyzed by dehydratases
- Requires pyridoxal phosphate
3. Deamidation
Amide-containing amino acids release ammonia.
Glutamine
Glutamine + H2O → Glutamate + NH3
- Catalyzed by glutaminase
Asparagine
- Asparagine → Aspartate + NH₃
4. Intestinal Production
- Intestinal bacteria degrade proteins and urea
- Large amount of ammonia formed in intestine
- Ammonia enters portal circulation and reaches liver
5. Purine and Pyrimidine Catabolism
Nitrogenous bases also produce ammonia during degradation.
Transport and Storage of NH₃
Because free ammonia is toxic, it is transported safely in non-toxic forms.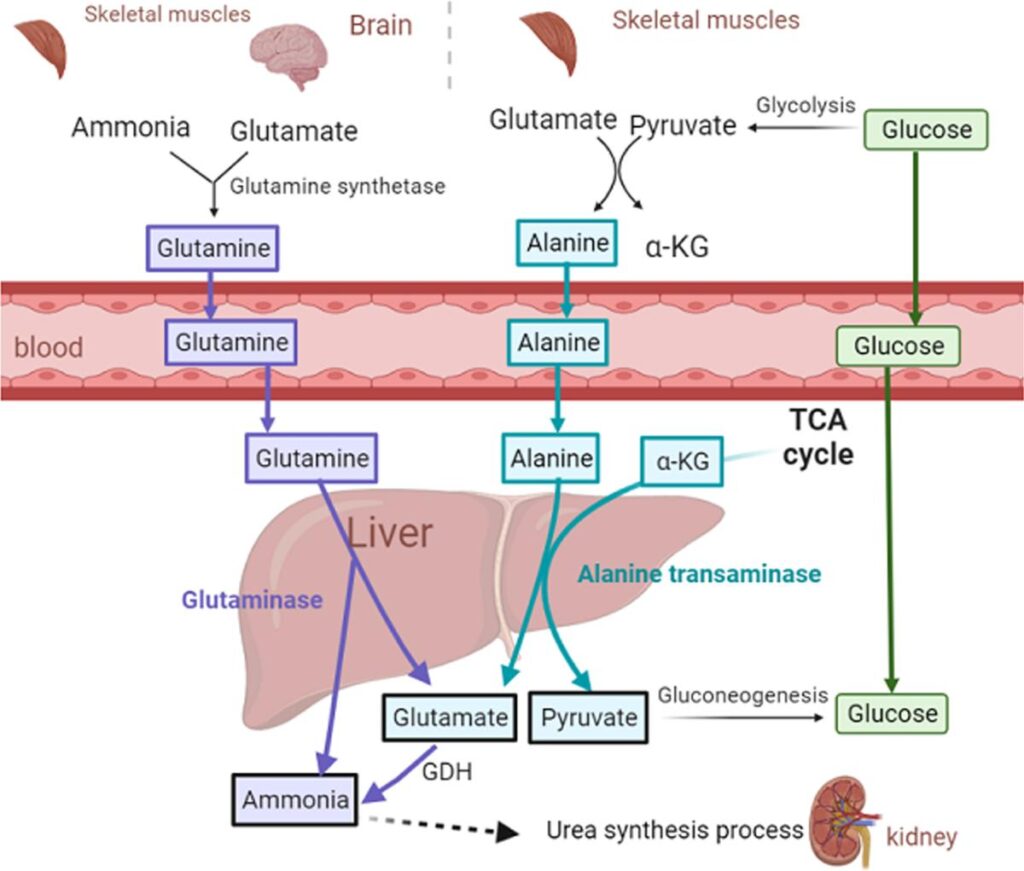
1. Transport as Glutamine
Glutamine is the major transport form of ammonia in blood.
Glutamate + NH3 + ATP → Glutamine
Enzyme:
- Glutamine synthetase
Features:
- Occurs in brain, muscle, and peripheral tissues
- Glutamine carries two nitrogen atoms
- Non-toxic storage form
In liver:
- Glutaminase releases ammonia again
2. Transport as Alanine
Important in skeletal muscle.
Pyruvate + Glutamate → Alanine + α-ketoglutarate
Features:
- Alanine transports amino nitrogen from muscle to liver
- Part of glucose-alanine cycle
- Liver converts alanine back to pyruvate
3. Temporary Storage
Ammonia can be temporarily stored in:
- Glutamine
- Glutamate
This prevents toxic accumulation.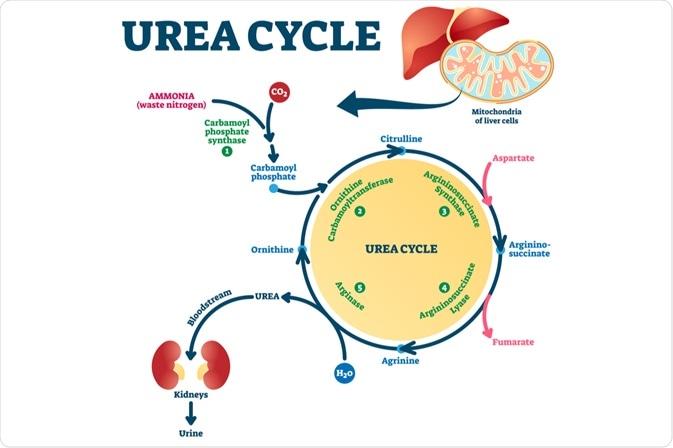
Functions of Ammonia
Although toxic in excess, ammonia has important metabolic functions.
Major functions
- Formation of urea in liver
- Synthesis of non-essential amino acids
- Used in synthesis of:
- Purines
- Pyrimidines
- Creatine
- Formation of glutamine
- In kidney, helps regulate acid-base balance
Kidney role
Ammonia binds hydrogen ions:
NH3 + H+ → NH4
- Ammonium is excreted in urine
- Helps remove acid from body
Disposal of Ammonia
The body must continuously remove ammonia.
1. Conversion into Urea (Major Pathway)
- Occurs in liver through urea cycle
- Major detoxification mechanism
2. Renal Excretion as Ammonium Ion
Kidney converts ammonia into ammonium ion.
Importance:
- Prevents acid accumulation
- Important during acidosis
3. Formation of Glutamine
Extrahepatic tissues temporarily detoxify ammonia by glutamine formation.
Toxicity of Ammonia
Ammonia is highly toxic, especially to brain tissue.
Mechanism of Toxicity
1. Depletion of α-ketoglutarate
Ammonia combines with α-ketoglutarate to form glutamate.
This reduces availability of α-ketoglutarate for TCA cycle.
2. Reduced ATP production
TCA cycle slows → ATP decreases.
3. Glutamine accumulation in brain
Excess glutamine causes osmotic swelling.
4. Cerebral edema
Brain swelling leads to neurological symptoms.
Clinical Manifestations
- Headache
- Irritability
- Vomiting
- Tremors
- Confusion
- Convulsions
- Coma
Causes of Hyperammonemia
- Severe liver disease
- Urea cycle enzyme defects
- Liver failure
- Neonatal metabolic disorders
Clinical Disorders
Hyperammonemia
- Increased blood ammonia
- Neurological emergency
Hepatic encephalopathy
- Seen in severe liver dysfunction